Material Selection for Medical Device Components: Matching Materials to Clinical Environments
The choice of materials for medical device components is a critical decision that directly impacts safety, performance, durability, and regulatory compliance. Different clinical environments—ranging from sterile operating rooms to implantable applications inside the human body—impose distinct demands on materials. Selecting the right material requires careful consideration of biological compatibility, mechanical properties, sterilization methods, and environmental exposure.
1. Implantable Devices: Biocompatibility Is Paramount
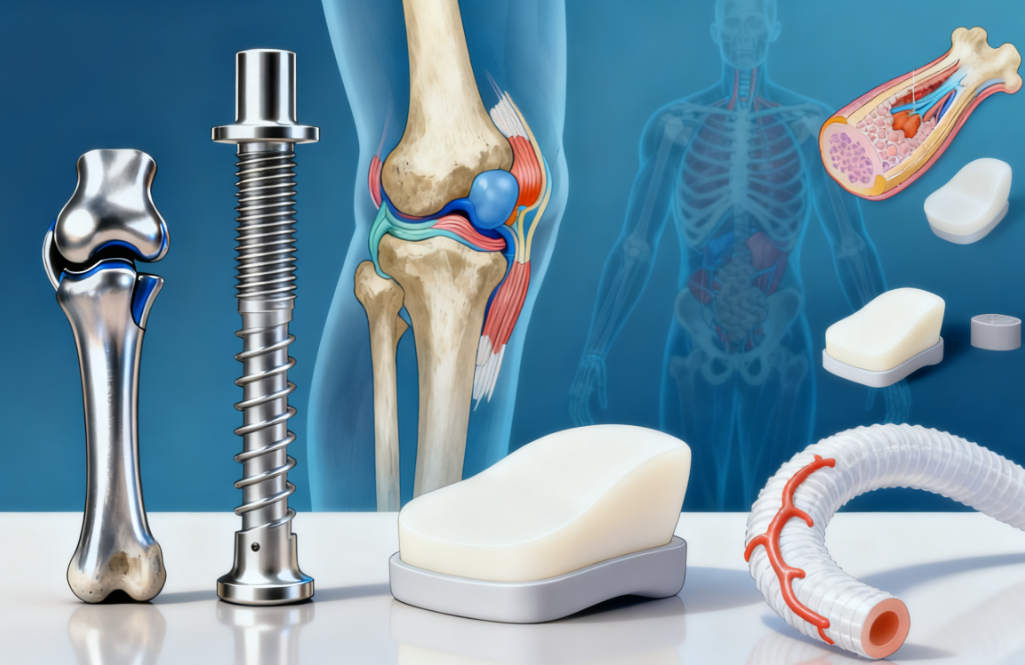
For devices intended to reside inside the human body—such as pacemakers, orthopedic implants, or vascular grafts—the material must exhibit exceptional biocompatibility, long-term stability, and mechanical resilience.
- Titanium and its alloys are widely used for bone implants due to their excellent strength-to-weight ratio, corrosion resistance, and ability to osseointegrate (bond with bone).
- Cobalt-chromium alloys offer high wear resistance, making them ideal for joint replacements.
- Medical-grade polymers like ultra-high-molecular-weight polyethylene (UHMWPE) are used in articulating surfaces of hip and knee implants for their low friction and wear properties.
- Polytetrafluoroethylene (PTFE) and polyurethane are common in soft-tissue implants and vascular grafts due to flexibility and blood compatibility.
Crucially, these materials must comply with ISO 10993 standards for biological evaluation and demonstrate no cytotoxicity, sensitization, or genotoxicity.
2. Surgical Instruments: Durability and Sterilizability
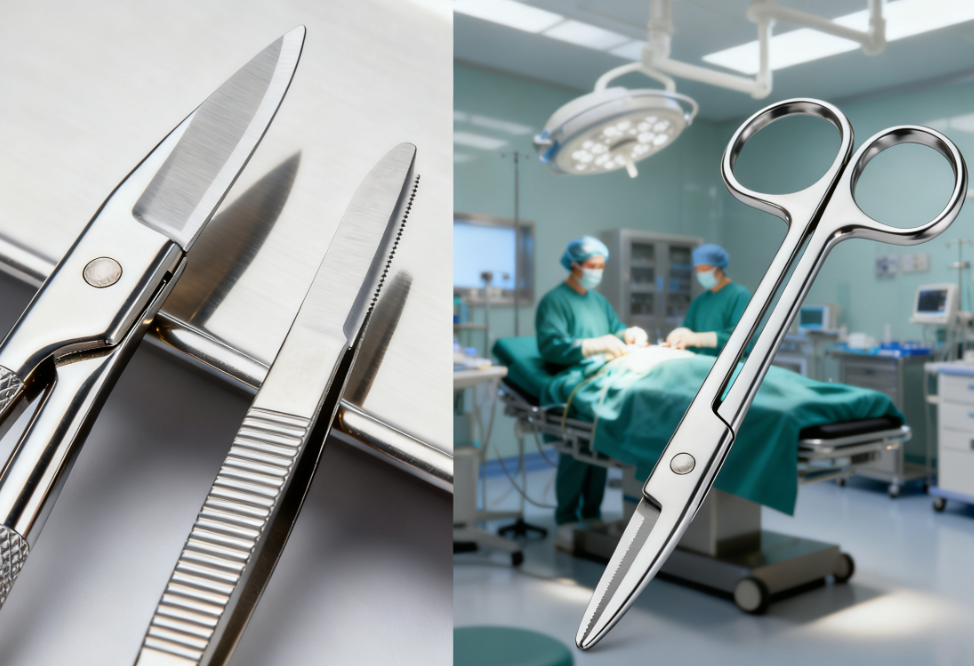
In operating rooms, instruments face repeated exposure to high-temperature steam sterilization, aggressive cleaning agents, and mechanical stress.
- Stainless steel (e.g., AISI 316L) remains the gold standard for reusable surgical tools due to its corrosion resistance, strength, and ability to withstand autoclaving.
- For single-use instruments, polycarbonate or polyetheretherketone (PEEK) may be chosen for their rigidity, clarity (for visualization), and compatibility with gamma or ethylene oxide (EtO) sterilization.
Materials here must maintain structural integrity over hundreds of sterilization cycles without degrading or leaching harmful substances.
3. Diagnostic and External-Use Devices: Safety and User Experience
Devices like ECG electrodes, blood pressure cuffs, or glucose monitors contact skin but are not implanted. Key considerations include skin compatibility, flexibility, and ease of manufacturing.
- Silicone and thermoplastic elastomers (TPEs) are favored for skin-contact parts due to softness, hypoallergenic properties, and comfort.
- Polystyrene and acrylonitrile butadiene styrene (ABS) are commonly used in housings for diagnostic equipment because they are easily molded, cost-effective, and can be made antimicrobial with additives.
These materials should resist degradation from alcohol wipes, UV light, and frequent handling while maintaining aesthetic quality.
4. Fluid-Contacting Components: Chemical Resistance and Purity
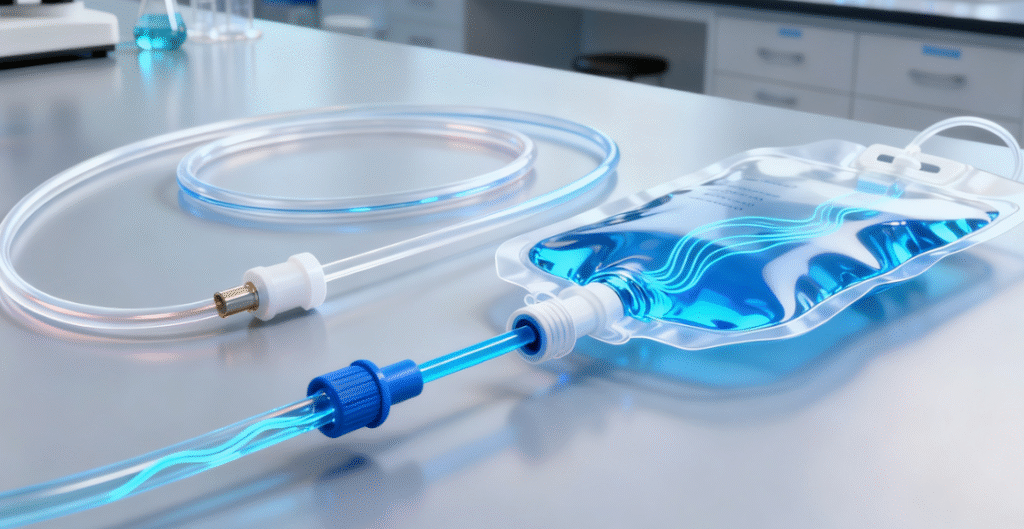
Tubing, catheters, IV bags, and fluid reservoirs must resist chemical attack from drugs, saline, or blood while preventing leaching.
- Polyvinyl chloride (PVC) with medical-grade plasticizers has been traditionally used, though concerns about phthalates have driven adoption of alternatives like thermoplastic polyurethanes (TPU) or polyolefins.
- Fluoropolymers (e.g., FEP, PFA) offer extreme chemical inertness and are used in high-purity applications like dialysis or drug delivery systems.
All materials must meet USP Class VI or equivalent standards for extractables and leachables.
5. Environmental Factors Across Settings
Beyond function, the clinical environment dictates additional requirements:
- Moisture-rich areas (e.g., endoscopy suites): require hydrolysis-resistant materials like PEEK or certain polyamides.
- Radiation-heavy zones (e.g., radiology): demand radiation-stable polymers that won’t yellow or embrittle.
- Cleanrooms and labs: favor non-shedding, low-particulate materials to maintain sterility.
Conclusion
There is no “one-size-fits-all” material in medical device manufacturing. Successful component design begins with a clear understanding of the intended use environment, duration of exposure, regulatory pathway, and end-user needs. By aligning material properties with clinical realities, engineers ensure devices are not only functional but also safe, reliable, and compliant across the diverse landscapes of modern healthcare.
Let me know if you’d like this tailored for a specific audience (e.g., engineers, regulators, students) or expanded into a longer white paper!